
(a) Monocyclic systems: Some monocyclic systems having π-electrons (obey Huckel’s rule) possess aromatic character.ī) Fused ring systems: The polynuclear hydrocarbons such as naphthalene, anthracene and phenanthrene are also aromatic according to Huckel’s rule.Īromatic ions: Some cyclic ions also exhibit aromatic character. Huckel's rule can be applied successfully to polycyclic compounds, annulenes and also other non-benzenoid compounds. According to this rule, a cyclic compound will behave as aromatic compound if it contains (4 n + 2) electrons, may be 0, 1, 2, 3 etc. (v) The most essential criteria for the aromatic character is that the compound must obey Huckel’s rule. (iv) The molecules have delocalised π electron cloud above and below the plane of the ring. (iii) They must resist addition reaction and take part in the electrophilic substitution reactions. (ii) Their molecular formulae suggest these compounds to be highly unsaturated due to the presence of one or more double bonds in the ring but they must behave as saturated compounds. (i) The compounds must be cyclic in nature and have flat planar structure. 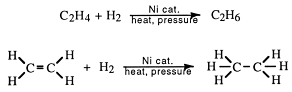
The compounds possessing aromatic character show the following characteristics:
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
